

Purchase tickets now for this celebratory reception and dinner at The Pendry, and be part of this high profile international event.
Read More
In this episode dedicated to the Australian Clinical Entrepreneur Program (AUSCEP), we meet Dr Sumedha Verma, a clinical psychologist and sleep expert, and the founder of Deep Sleep Health.
Listen to Podcast
180+ delegates, 21 exhibitors, 7 Pathway to Market pitches and a packed program focused on tackling one question…
Read More
Read the new case by BiVACOR, an Australian-born medical device company, who are developing a next-generation titanium Total Artificial Heart (TAH) to restore the lives of people with end-stage heart failure.
Read More
Read a new case study by University of Sydney spin-out Endo Axiom who are using nanotechnology to develop smart oral insulin in a pill with support from MTPConnect’s Targeted Translation Research Accelerator (TTRA).
Read More
Read the case study by Perth-based SME VitalTrace who is developing DevliAssure, real-time Fetal monitoring for safer childbirth with funding support through MTPConnect’s Clinical Translation and Commercialisation Medtech (CTCM) program.
Read More
Investment of $1 million from MTPConnect’s Clinical Translation and Commercialisation Medtech (CTCM) program has helped propel VividWhite’s novel surgical implant, VividFlo, towards becoming an improved long-term treatment option for people with glaucoma.
Read More
Case Study: Ventora Medical is developing an Airway Pressure Monitor that delivers non-invasive respiratory treatment for premature babies requiring neonatal intensive care support, with support from our CTCM program.
Read More
Atrial fibrillation (AF) is a most common heart rhythm disorder that can be treated with catheter ablation. CathRx, Australia’s only onshore designer and manufacturer of electrophysiology catheters, is developing an innovative pulsed field ablation technology to improve treatment with the help of MTPConnect’s CTCM accelerator.
Read More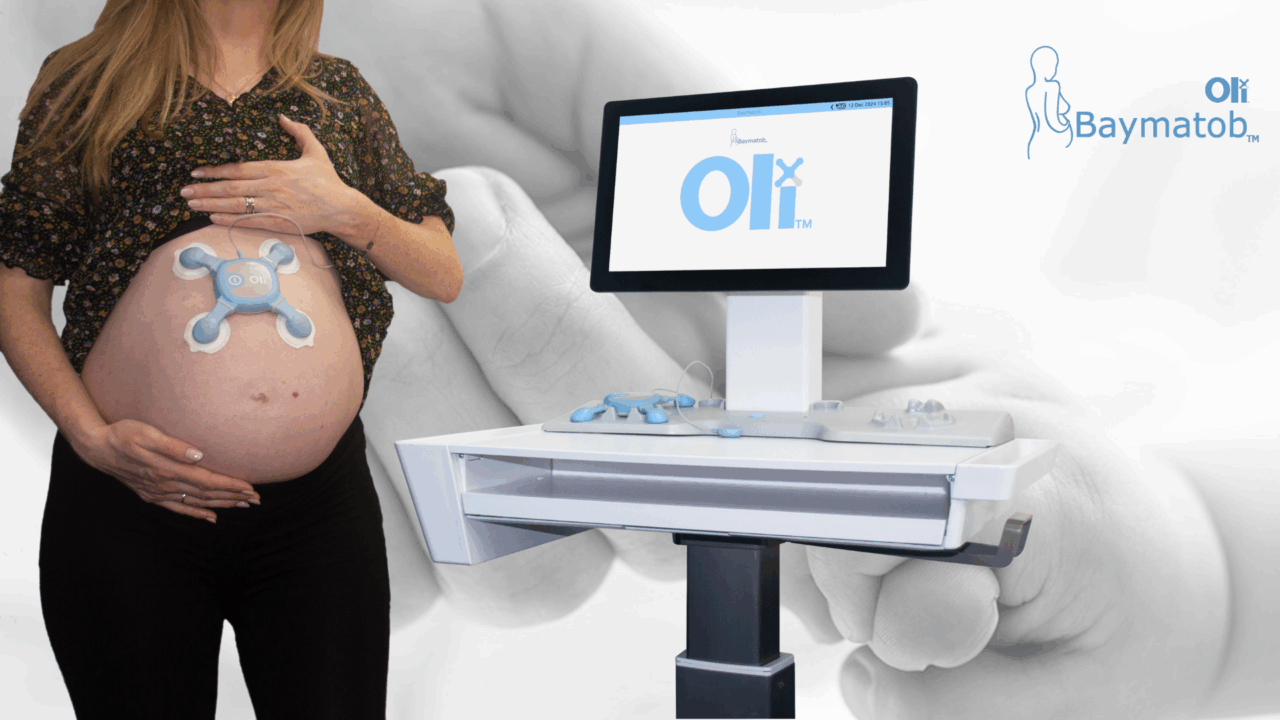
The AUSCEP team caught up with AUSCEP mentor and medtech commercialisation expert, Tara Croft, to learn about Oli – a groundbreaking new medical device that is aiming to make pregnancy and childbirth safer and better for women.
Read More
Placing central venous catheters in newborns is one of the most delicate and error-prone procedures – Navi Medical Technologies is changing that with support from CTCM program, a MRFF initiative delivered by MTPConnect.
Read More