The Clinical Translation and Commercialisation Medtech (CTCM) program, designed and delivered by MTPConnect, is offered under the 2020 Early Stage Translation and Commercialisation Support Grant of the Medical Research Future Fund’s Medical Research Commercialisation Initiative.
The $19.75 million program identifies and nurtures high-quality medical device projects that have commercial potential and support their translation through early clinical trials. The program is focused on Australian small to medium-sized enterprises (SMEs) and aims to boost commercialisation of home-grown medical products.
Projects are focused on the development of a medical device for human use, the purpose of which is to diagnose, prevent, monitor, treat or alleviate a disease or injury, or modify or monitor anatomy or physiological functions of the body. Funding has been awarded to projects where the device’s capability has already been validated with research and/or preclinical studies.
By providing funding of between $250,000 and $1.5 million per project across two funding rounds (FY2022 and FY2023), the CTCM Program is:
- Delivering consultation and commercialisation advice to guide project development and assessment
- Facilitating access to broader NCRIS and other critical engineering, fabrication and prototyping facilities to accelerate translation of early-stage discoveries
- Emphasising collaboration, partnering and consultation to nurture the next generation of health and medical research innovators and provide ongoing SME education
- Employing a process of continuous evaluation, based on established commercial principles, to optimise the potential for project success and maximise return on investment.
By supporting the development of innovative medical devices, the CTCM program aims to improve the health and wellbeing of Australians, while also helping projects to generate commercial returns and creating high-paying jobs in the medical products sector.
MTPConnect is delivering the program in partnership with our highly esteemed medical technology commercialisation, education and infrastructure partners: Medical Technology Association of Australia (MTAA), Medical Device Partnering Program (MDPP), Cicada Innovations, the BridgeTech Program and Therapeutic Innovation Australia (TIA).
News resources
Visit our News page and select CTCM to discover news stories, case studies, podcasts and reports!
Driving Australian Medtech Innovation Forward
As MTPConnect’s Clinical Translation Commercialisation Medtech (CTCM) Program moves to completion, a new report showcases the exciting new technologies being developed by some of Australia’s most innovative emerging medtech companies with support from the program.
Established in 2021, with support from the Medical Research Future Fund (MRFF), the program’s unique accelerator model designed and delivered by MTPConnect aims to get technologies to patients quicker with the focus on enabling early-stage clinical trials, providing wrap-around support and deep technical and commercial expertise, all with a view to encouraging company growth.
The impact is outlined in the report Driving Australian Medtech Innovation Success, was released to coincide with MTPConnect’s Accelerator Showcase in Melbourne celebrating homegrown innovations and opened by the Minister for Health, Ageing and Disability the Hon Mark Butler MP.
Download CTCM Impact ReportResources
Case Study

VividWhite set to transform surgical glaucoma management and vision care
Investment of $1 million from MTPConnect’s Clinical Translation and Commercialisation Medtech (CTCM) program has helped propel VividWhite’s novel surgical implant, VividFlo, towards becoming an improved long-term treatment option for people with glaucoma.
Read MoreCase Study
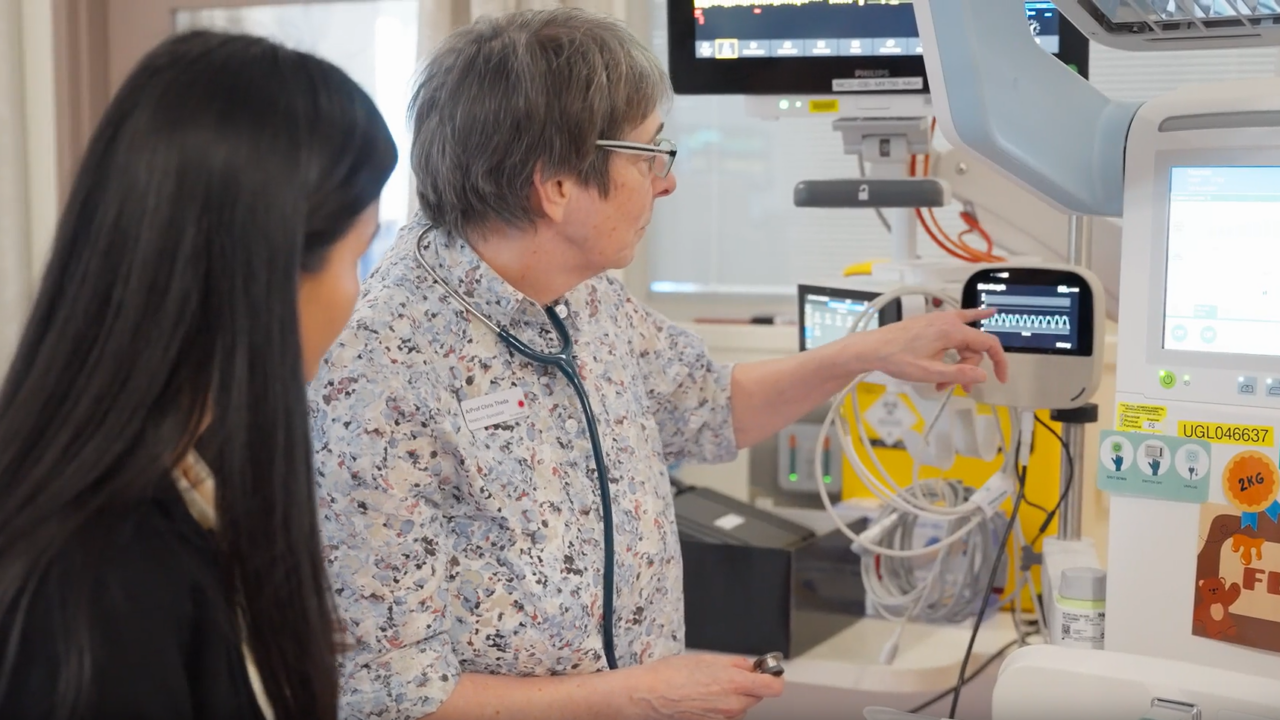
Ventora Medical – Improving respiratory support for newborns
Case Study: Ventora Medical is developing an Airway Pressure Monitor that delivers non-invasive respiratory treatment for premature babies requiring neonatal intensive care support, with support from our CTCM program.
Read MoreNews
Case Study

CathRx: Transforming atrial fibrillation treatment with innovative pulsed field ablation technology
Atrial fibrillation (AF) is a most common heart rhythm disorder that can be treated with catheter ablation. CathRx, Australia’s only onshore designer and manufacturer of electrophysiology catheters, is developing an innovative pulsed field ablation technology to improve treatment with the help of MTPConnect’s CTCM accelerator.
Read MoreCase Study
Podcasts

175. VitalTrace: Revolutionising Childbirth Monitoring to Keep Mothers and Babies Safe
The Clinical Translation and Commercialisation Medtech (CTCM) program aims to boost commercialisation of home-grown medical products. It is delivered by …
Listen to Podcast